
Genentech, a member of the Roche Group has announced that the U.S. Food and Drug Administration (FDA) has granted Breakt...

Merck, known as MSD outside the United States and Canada, has announced that the U.S. Food and Drug Administration (FDA)...
Certara®, the global model-informed drug development and decision support leader, on 12 Sep 2019, announced the laun...

Chugai Pharmaceutical Co., Ltd. (TOKYO:4519) announced that the results from SAkuraStar Study were presented at the...

Hansoh Pharmaceutical Group Company Limited (“Hansoh Pharma”), a leading biopharmaceutical company in China,...

Daiichi Sankyo Company, Limited (hereafter, Daiichi Sankyo) has announced the submission of a New Drug Application...

Invicro , a member of the Konica Minolta Group and a leading provider of biomarkers for imaging, core lab services and s...
THC Global Group Limited (THC Global or the Company) has announced that its Southport Facility was opened by the Federal...

AstraZeneca has announced that the US Food and Drug Administration (FDA) has granted Fast Track designation for the deve...

Cantargia AB and BioWa Inc. have signed an extension of the license agreement around the BioWa proprietary POTELLIGENTTe...

Voronoi, drug discovery and development biotech company in Korea has announced that it has, after an extensive evaluatio...
Innovent Biologics, Inc., a world-class biopharmaceutical company that develops and commercializes high-quality medicine...

The U.S. Food and Drug Administration, on 19 August 2019, approved Xenleta (lefamulin) to treat adults with community-ac...

Pretomanid, a novel compound developed by the non-profit organization TB Alliance, was approved by the U.S. Food & D...

Genentech, a Roche’s subsidiary announced that the US Food and Drug Administration (FDA) has approved Rozlytrek&tr...

Certara®, the global model-informed drug development and decision support leader, on 30 July 2019, announced the lau...

Bayer AG and BlueRock Therapeutics hasannounced an agreement under which Bayer will fully acquire BlueRock Therapeutics,...

Sun Pharmaceutical Industries Ltd. has announced that one of its wholly owned subsidiaries has filed an application for ...

Pfizer Inc. announced the successful completion of its acquisition of Array BioPharma Inc., advancing breakthrough...

3SBio Inc., a leading biopharmaceutical company in the People's Republic of China (PRC) has announced that an applicatio...

AstraZeneca has announced that data from two Phase III trials evaluating the efficacy and safety of roxadustat in China....

Japan headquartered Sosei Group Corporation has entered into a multi-target research collaboration and license agreement...

Japanese pharmaceutical firm Eisai Co.,Ltd. has announced that it has entered into a collaboration research agreement wi...

CEL-SCI Corporation (NYSE American: CVM) announced that one of its key collaborators from Rush University Medi...

Gilead Sciences, Inc., on 24 June 2019, announced that the Singapore Health Sciences Authority (HAS) has approved Vosevi...

Gilead Sciences and Nurix Therapeutics, Inc., have announced a global strategic collaboration to discover, dev...

Zydus’ Nesher Pharmaceuticals, a subsidiary of Zydus Pharmaceuticals USA has received the final approval from the ...
Acucela Inc. (“Acucela”), a clinical-stage ophthalmology company and wholly-owned subsidiary of Kubota Pharm...

Sosei Group Corporation and its strategic alliance partner Pfizer announce that a new clinical candidate from their mult...

CStone Pharmaceuticals, on 9 June 2019, announced its global clinical collaboration with China focus with Baye...

Rykindo® (LY03004), Luye Pharma's innovative, independently developed Extended-Release Microspheres f...

Sihuan Pharmaceutical Holdings Group Ltd., the largest cardio-cerebral vascular (CCV) drug manufacturer in China's&...
BGN Technologies, the technology transfer company of Ben-Gurion University (BGU), introduces a novel drug comb...

Gilead Sciences, Inc. has recently announced that with effect from 1 May 2019, Vemlidy (tenofovir alafenamide, TAF)...

ADC Therapeutics, an oncology drug discovery and development company that specializes in the development of proprietary ...
Roche has declared that Emicizumab (Hemlibra®) has been approved in India for Hemophilia A with factor VIII in...

Eisai Co., Ltd. is a leading global research and development-based pharmaceutical company headquartered in Japan. The co...

Takeda Pharmaceutical Company Limited announced that the European Medicines Agency (EMA) has accepted a Marketing Author...

Standigm, a company using artificial intelligence (AI) technology for drug discovery and development, announced it has r...

StrideBio, Inc, a leading developer of novel adeno-associated viral (AAV) based gene therapies, recently announced the s...

HitGen has announced that the company has entered into a research collaboration with Almirall to identify novel small mo...

BioInvent International AB, focused on the discovery and development of novel and first-in-class immuno-regulatory antib...

Roche announced that the European Commission has approved MabThera® (rituximab) for the treatment of adults with mod...

Ono Pharmaceutical Co., Ltd., a Japanese pharmaceutical research and development company, and twoXAR, Inc., a US based a...

The Janssen Pharmaceutical Companies of Johnson & Johnson announced the submission of a supplemental Biologics Licen...

The U.S. Food and Drug Administration has approved Spravato (esketamine) nasal spray, in conjunction with an oral antide...

Glenmark Pharmaceuticals has announced the presentation of a new post hoc analysis of data from a Phase 2a, proof-of-con...
Merck and Pfizer Inc. has announced that the US Food and Drug Administration (FDA) has accepted for Priority Review...

Teva Pharmaceutical Industries has announced the launch of a generic version of Sabril®1 (vigabatrin) tablets, ...
Perrigo Company plc has announced it has received final approval from the U.S. Food and Drug Administration for its AB r...
BioLineRx Ltd., an Israeli clinical-stage biopharmaceutical company focused on oncology, announced that the U.S. Food an...

Reaction Biology Corporation ("RBC") and ChinaEquity Group ("CEG") have announced the conclusion of a stock exchange tra...

I-Mab Biopharma (I-Mab), a China-based clinical stage biopharmaceutical company exclusively focused on the developm...

Eli Lilly and Companyhas announced that USFDA has granted approval for a new indication for ALIMTA® (pemetrexed...

Neurocrine Biosciences and Voyager Therapeutics has announced the formation of a strategic collaboration focused on the ...

Pfizer and Eli Lilly have announced positive top-line results from a Phase 3 study evaluating tanezumab 2.5 mg or 5 mg i...

The Janssen Pharmaceutical Companies of Johnson & Johnson has announced the U.S. Food and Drug Administration (FDA) ...

Commercial stage biopharma company, Basilea Pharmaceutica has announced that it entered into a collaboration with Roche ...

UK based biopharma company, Wren Therapeutics focused on drug discovery and development for protein misfolding diseases ...

AbbVie, a research-based global biopharmaceutical company has announced an update on the Phase 3 RESOLVE trial (PCYC-113...

Spero Therapeutics, Inc., a multi-asset clinical-stage biopharmaceutical company focused on identifying, developing and ...

The Janssen Pharmaceutical Companies of Johnson & Johnson has announced the submission of a Group Type II Variation ...

Denali Therapeutics Inc., a biopharmaceutical company developing a broad portfolio of therapeutic candidates for neurode...

Amgen and UCB announced that the Japanese Ministry of Health, Labor and Welfare has granted a marketing authorizat...

Exelixis, Inc. has announced that its partner Daiichi Sankyo Company, Limited received approval from the Japanese Minist...

Cellular Biomedicine Group Inc., a biopharmaceutical firm engaged in the development of immunotherapies for cancer and s...

Oculis SA, a clinical-stage biopharmaceutical company developing next-generation topical ophthalmic treatments recently ...

Biopharma company, AbbVie and Biotech company, Tizona Therapeutics has announced that they have entered into a global, s...

Bristol-Myers Squibb Company has announced the U.S. Food and Drug Administration (FDA) has expanded the indication for&n...

AstraZeneca has announced that the Phase III OLYMPUS and ROCKIES trials for roxadustat each met their primary efficacy e...

Pharma major Lupin announced that it has received approval for its Clobazam Oral Suspension, 2.5 mg/mL from the United S...

Teva Pharmaceutical Industries Ltd., today announced the launch of a generic version of Elidel®1 (pimecrolimus)...

BeiGene, Ltd., a commercial-stage biopharmaceutical company focused on developing and commercializing innovative molecul...

Chugai Pharma has announced today that it has obtained regulatory approval from the Ministry of Health, Labour and Welfa...

Cannabics Pharmaceuticals Inc., a world leader in personalized cannabinoid medicine focused on cancer and its side effec...

I-Mab Biopharma (I-Mab), a China-based clinical-stage biopharmaceutical company exclusively focused on the developm...

Roivant Sciences has announced that it has entered into collaboration with Daiichi Sankyo Company, Limited (hereafter, D...

Biopharma company Lexicon Pharmaceuticals, Inc. has announced positive topline results from its Phase 1a study of LX9211...

Bridge Biotherapeutics Inc., a clinical stage biotech company headquartered in Seongnam, South Korea, has signed a licen...

Singapore – China's National Medical Products Administration approves FibroGen's roxadustat, a hypoxia-i...

UK-based Avacta Group plc , the developer of Affimer biotherapeutics and reagents and Korea's LG Chem Life Science...
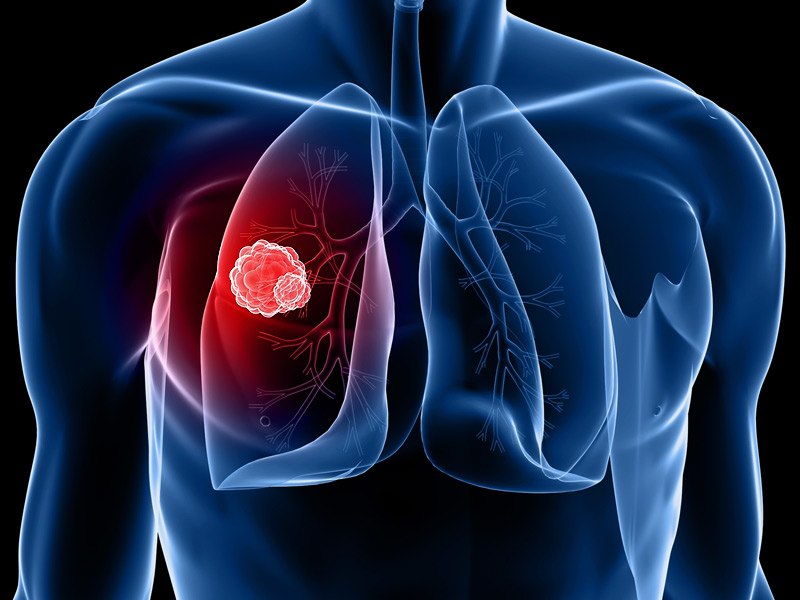
Innovent Biologics, Inc. (Innovent), a world-class China-based biopharmaceutical company that develops and commerci...

Multiple myeloma patients from Australia will have world-first access to a new first-in-class drug developed to treat th...

TauRx, a leader in neurodegenerative disease research, announced that the US Food and Drug Administration (FDA) has gran...

Diamyd Medical is dedicated to finding a cure for diabetes and other serious inflammatory diseases through pharmaceutica...
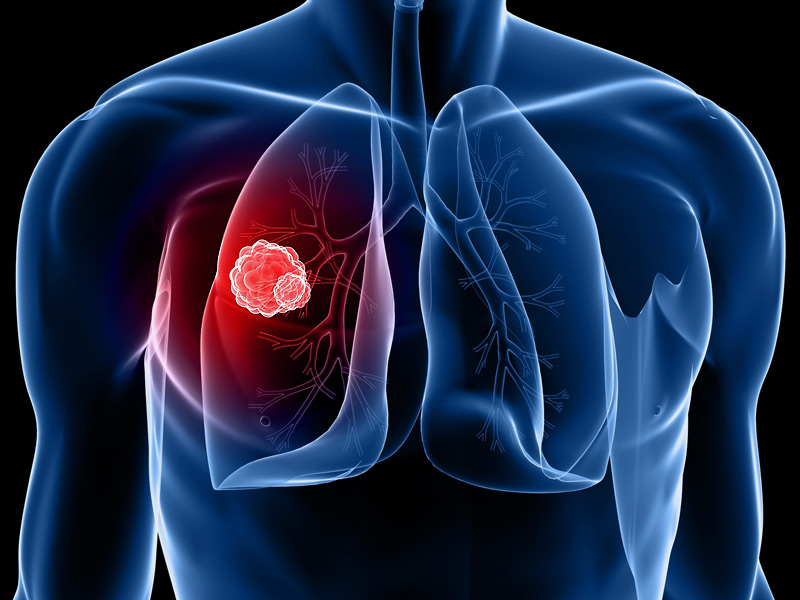
Takeda Pharmaceutical Company Limited announced that the European Commission (EC) granted marketing authorization for AL...

HitGen Ltd has announced that the company has entered into a drug discovery research collaboration with Biogen to identi...

Singapore – Novartis announced that the Health Sciences Authority (HSA) has approved Aimovig (erenumab) ...

OBI Pharma, Inc., a Taiwan biopharma company, has announced that the U.S. Food and Drug Administration (FDA) h...

Singapore - Exscientia, the company leading the use of Artificial Intelligence (AI) in drug discovery, announces that is...

MEI Pharma, Inc. and Kyowa Hakko Kirin Co., Ltd. (KHK), have announced the execution of a license agreement granting KHK...

Singapore – The Janssen Pharmaceutical Companies of Johnson & Johnson announced that the U.S. Food and D...

Singapore – Halozyme has out-licensed its ENHANZE drug delivery technology to Roche for the exclusi...

Singapore – A non-opioid drug developed by Pfizer and Eli Lilly met the main go...

Singapore - The FDA approves Regeneron Pharmaceuticals and development partner Sanofi's DUPIXENT (dupilumab) a...

Singapore - Results from a Phase 3 clinical trial, IMpassion130, evaluating Roche unit Genentech's TECENTRIQ (...

Singapore - MAZ World and ZEAL Cosmetics have confirmed that Ostrich Antibodies are effective in neutralising all four s...

Strides Pharma Science (Strides) on Saturday announced that its global arm Strides Pharma Global Pte Singapore has recei...

A joint research group centered around Professor Hideyuki Okano and Associate Professor Jun Kohyama, Department of Physi...

Singapore - Axsome Therapeutics, a clinical-stage biopharmaceutical company developing novel therapies for the mana...



