Photo Credit: Freepik
Sona Nanotech Inc., a developer of rapid, point-of-care diagnostic tests, announced on 2 July 2020 that its rapid detection, COVID-19 antigen test’s laboratory validation studies of performance levels have resulted in a test sensitivity of 96%, test specificity of 96% and a Limit of Detection (“LOD”) of 2.1 x 102 TCID50. Sales of the tests will now be permitted under a ‘research use only’ label until full regulatory authority is granted, in relevant territories, at which time the ‘research use only’ label requirement would be lifted, as discussed below. Technology transfer to manufacturers is currently underway to produce tests to meet current and expected demand. The Company will provide an update on sales progress and manufacturing delivery timetables in the coming weeks.
MRIGlobal, using live COVID-19 viral cultures, determined the test to have a limit of detection of 2.1 x 102 TCID50 which corresponds to an ability to detect the virus in patients with ‘low’ viral loads in 10-15 minutes, as compared to RT-PCR testing which typically takes 24-48 hours to detect the virus. Limit of detection is the minimum amount of target microorganisms that can be reliably detected under optimal conditions and is an essential step in determining the sensitivity of any assay. Current studies show positive COVID-19 patients presenting symptoms have viral loads in the 104 – 106 range.
Validation studies were also conducted in-house to assess potential clinical performance of the test using 30 nasopharyngeal samples from healthy individuals who were presumed negative for COVID-19. Results from the study generated a specificity of 96% (29/30) and a sensitivity of 96% (28/29). All specimen samples tested generated negative results, except for one, generating the above result of 96%. To generate the sensitivity data, the remnants of each negative sample were spiked with gamma irradiated COVID-19 virus and the tests rerun to determine the positive results, generating the above result of 96%.
Following consultation with MRIGlobal and the FDA, Sona will enter into independent clinical, in-field evaluation studies to generate the data to support its analytical and clinical data as part of the submission it will make to Health Canada and the FDA for emergency use authorization (“EUA”) approval. In-field collection of a minimum of 30 confirmed negative and 30 confirmed positive specimens and the associated data analysis is expected to be completed while technology transfer to manufacturers is still underway. To that end, the Company has engaged with a contract research organization (“CRO”) based in the U.S. to conduct one such study and a university-affiliated laboratory outside of the U.S. to conduct a second. The Company has been informed that the results of these field studies should be provided by the end of July, at which time it intends to make final submissions to regulatory authorities in multiple jurisdictions. During this time, technology transfer will continue and quality assurance manufacturing batches are expected to be run with manufacturing partners. The Company is committed to maintaining ongoing evaluations of its test in order to understand its performance in a wide range of testing environments.
Darren Rowles, CEO of Sona Nanotech, commented, “These excellent performance results are underpinned by our unique nanorod technology and completes a further milestone achieved for Sona along our path to bring a quality rapid test to market at scale. This will allow expansion of testing by governments, help ease the burden on healthcare systems, keep healthcare workers safe by allowing them to know their status on a daily basis and assist in softening restrictions by providing a quick and simple means to screen individuals. Our team have worked tirelessly to bring this product to this point in a record time and my thanks goes out to all involved in progressing the test this far.”
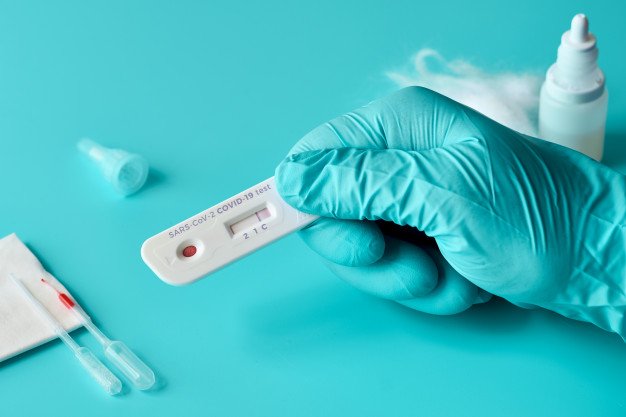
Rapid, point-of-care, antigen tests uses a nasopharyngeal swab to collect samples, which are then placed in a proprietary reagent solution and added to the sample port of the lateral flow test cassette. Blue colored lines will appear to indicate either a positive (2 lines) or negative result (1 line) within minutes.
The Company cautions that its COVID-19 rapid antigen test is not yet approved by the FDA or other regulatory bodies and will update the market as appropriate.




