10.. What kind of partnerships will APAC CROs see in the next 3 years

The fifth edition of the annual BioSpectrum Asia Pacific clinical research organization (CRO) Survey, which is conducted in association with CyberMedia Research (CMR), yet again identifies the latest trends, issues, perspectives and insights regarding the CRO industry in Asia Pacific (APAC).
While the 2013 survey revealed many hindrances facing the industry, including lack of optimum regulatory efficiency, training of personnel, limited development partnerships between CROs and pharma firms; it also reveals many promising findings, including high willingness to participate in clinical trials among countries like China, India, Australia, Korea, Taiwan, Malaysia, Philippines and Indonesia. Furthermore, the CRO sector in APAC is witnessing a major boom leveraging on the growth of logistics and supply chain management in the region.
An overwhelming majority of the survey respondents of the fifth BioSpectrum Asia-CMR Asia Pacific Annual Survey of the Clinical and Contract Research Organizations 2013, continue to repose their faith in the Asia Pacific market to increase further.
Speedy marketing to determine partnerships with pharma MNCs
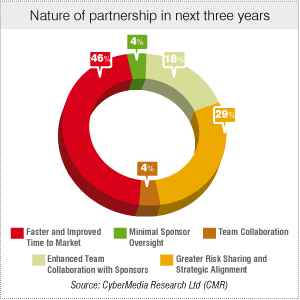
As many as 46 percent of the survey respondents indicate that faster and improved time to market will determine the nature of their partnership with pharma MNCs in the next three years. Of the survey respondents, 29 percent believe in greater risk sharing and strategic alignment for coming years.




