Perhaps even more significant is how important the industry has proven to be to national security, with the comparative strengths and weaknesses of countries seemingly amplified by the effects of the pandemic.
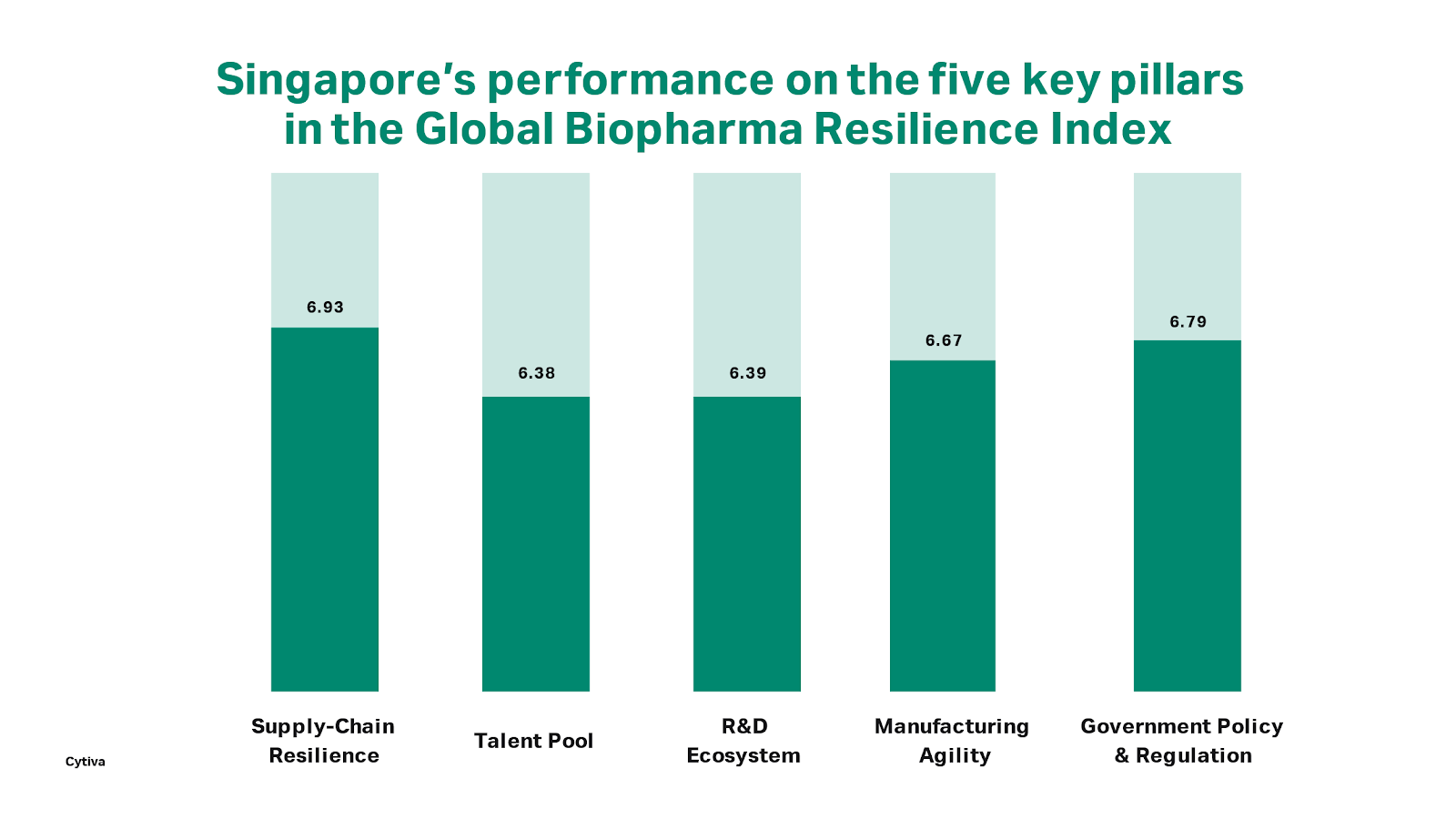
Cytiva recently released the results of its Global Biopharma Resilience Index, developed in partnership with Financial Times company Longitude, which tracks the resilience of the biopharma ecosystem in 20 countries by examining five areas of relevance: supply chain resilience, access to talent, strength of the R&D ecosystem, quality and agility of manufacturing capacity, and effectiveness of government policy in supporting the industry.
As one of the 20 countries covered in the research, Singapore’s standing in the Index, as expected, high in comparison with other ASEAN and Asian countries. But when compared against other developed nations with similar GNIs, such as the US and Australia, Singapore’s performance is notably poorer. While the results are not a huge cause for alarm, they do present a good opportunity to take a look at how the nation can develop further. In the process, they also left me asking myself the role I believe Singapore should be playing in the regional, or even global biotechnology roadmap.
Singapore’s advantages when it comes to economic development are so well-known that they’re virtually synonymous with the country itself: the ability to attract world-class talent, a bridge between east and west, and the openness to embrace new technologies. These are the reasons that Cytiva has been able to grow such a strong presence in Singapore, covering everything from our commercial operations and training facilities to our regional distribution centre for Asia-Pacific and our manufacturing site.
When we look deeper into the results of our research, we can spot areas where Singapore regulators can be more involved, for example developing a biopharma ecosystem. This includes developing a talent pool for the industry, especially given the large demand for GMP certified manufacturing talent not only in Singapore but in other ASEAN countries. I believe Singapore can also enhance its role in fostering greater collaboration between universities and biotech players. This is particularly important given that one in four respondents in the survey reported it as a “substantial challenge” to form such partnerships in Singapore, compared with just one in eight in the UK or the US.
Working with organizations and institutions throughout the biopharma value chain is among Cytiva’s core strengths, and we know that building a strong, collaborative ecosystem not only requires commitment from the local government, but also from industry and academia. Some of our recent success stories in driving scientific innovation through collaboration here in Singapore include our work with the National Cancer Centre Singapore on cell and gene therapy and with the Bioprocessing Technology Institute (BTI) on bioprocessing.
We also recently opened the Cytiva Experience and Learning Lab in Singapore, which provides hands-on training and leading industry know-how to researchers and biomanufacturers, supporting them in bringing their innovations through development and to market more efficiently and effectively. With 70 training sessions planned between now and the end of the year, the Lab will act as a platform for idea exchange and provide access to the latest technologies in biopharma manufacturing for those who could really benefit from them.
Elsewhere in the Index, no other country performs quite as well as Singapore when it comes to tax policies, and its efficient business environment and policies aimed at the development of biotech have attracted leading global biologics manufacturers and many start-ups. However much more is still needed if we are to really position Singapore as a global hub for biopharma. There is a need to create and strengthen the ecosystem to better support biotech start-ups in their journey, facilitate their access to networks and funding, and establish a vibrant environment with both local and global companies. This is not the sole responsibility of any single organization, but for all of us in this industry.
Singapore’s potential is there for the taking – it’s just up to us to make it happen.
By Rajan Sankaran, General Manager, ASEAN, Cytiva




