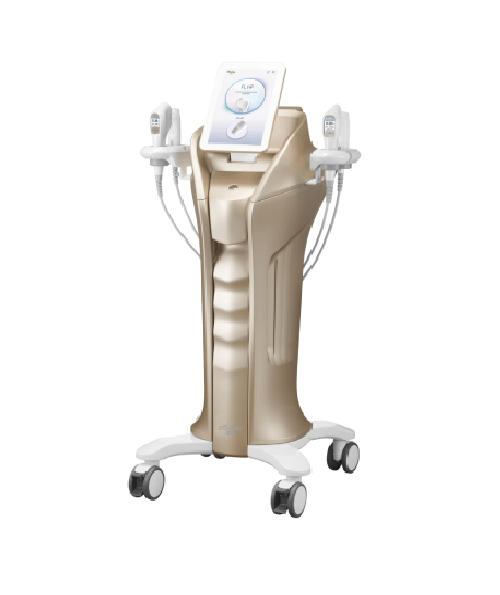
South Korea-based HIRONIC, a leading provider of aesthetic medical devices, has obtained certification from the Indonesian Ministry of Health (KEMENKES RI) for its flagship product, New Doublo 2.0—a state-of-the-art device combining 4th-generation High-Intensity Focused Ultrasound (HIFU) and Radiofrequency (RF) technologies in a single treatment platform.
This approval officially opens the door for exports to one of Southeast Asia's fastest-growing and largest aesthetic markets.
New Doublo 2.0 stands out as a comprehensive device that integrates HIFU, RF, and microneedling technologies—allowing practitioners to perform a wide range of aesthetic treatments using one platform. In the Indonesian market, rising demand for enhanced treatment efficacy and diversified procedure options has fueled interest in premium devices. Beauty clinics, in particular, are seeking differentiated brands to strengthen their positioning.
With the acquisition of the Indonesian certification, HIRONIC expects New Doublo 2.0 to establish itself as a flagship solution for premium lifting and multi-layer skin rejuvenation in the local aesthetic and medical sectors. Internationally recognized for its technological excellence and safety, New Doublo 2.0 will now target top-tier clinics across major Indonesian cities.
Following New Doublo 2.0, the company recently unveiled SYNERJET PRO, its latest skinbooster delivery system, reaffirming its commitment to innovation and market leadership in the global aesthetic device industry.




