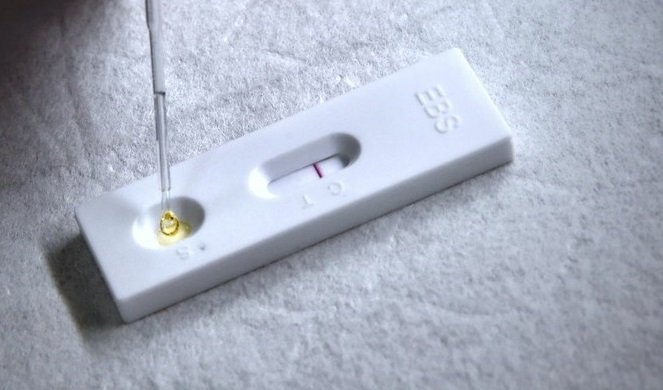
Photo credit: EBS
Taiwan's local biomedical company has developed a rapid antibody test kit for COVID-19 that can deliver results in just 10 minutes. The kit has passed clinical trials and is ready for mass production.
The rapid test kit for the detection of antibodies of SARS-CoV-2 has been validated by a comprehensive clinical study conducted by National Taiwan University Hospital (NTUH). The test boasts 100 per cent sensitivity and 95 per cent specificity, said Cooky Chen, chairman of Excelsior Bio-System, Inc., at an event for the presentation of the results.
The kit accessories and method functions similar to a pregnancy test kit, which uses two red lines to indicate results. Two lines indicate that the test is positive for SARS-CoV-2 antibodies. The test kit was successful in detecting antibodies in 13 COVID-19 patients, says project manager Hsieh Szu-min, an infectious disease specialist from NTUH.
Through the clinical trials, the hospital provides references to the company, including methods of execution and conducting of the testing, and offers SARS-CoV-2-positive patient samples and negative samples it has collected during the pandemic for a double-blind study to evaluate the sensitivity and specificity of the test, Hsieh said.
The kit screens both symptomatic or asymptomatic patients, however, as patients do not produce antibodies to the virus during the initial stages of infection, the antibody test can miss the early stages of infection and should be used in conjunction with the COVID-19 antigen rapid testing technology, said Lee Yu-chun, the head of the company's testing kit development division.
The kit was sent to the Health Ministry on May 5 for approval and is ready to be put into mass production, Chen said, adding that the company plans to produce 400,000 tests per month and introduce fully automated packaging lines for the product in the second half of this year to meet strong demand at home and from abroad amid the global pandemic.




