Brightonix Imaging, a South Korea-based startup, has announced that its flagship product, the PHAROS PET Scanner, has received US FDA clearance for commercial distribution in the United States (US).
This milestone marks a new era of precision and efficiency in nuclear imaging and positions Brightonix Imaging at the forefront of medical innovation.
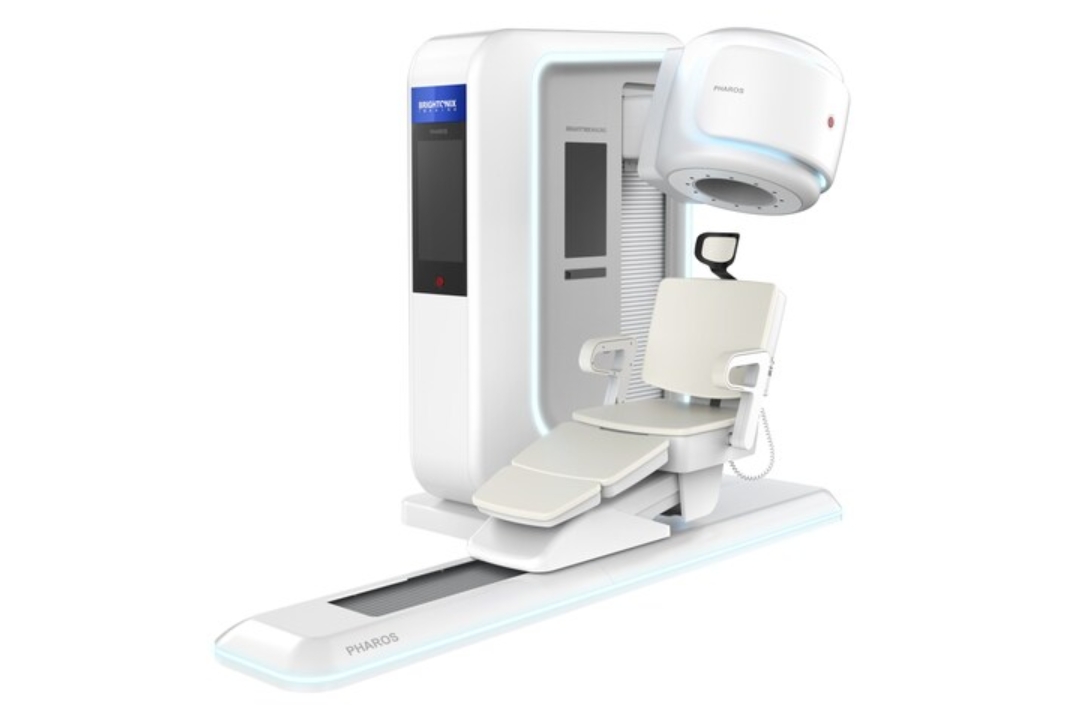
The PHAROS PET is a state-of-the art clinical positron emission tomography (PET) system designed to deliver exceptional image quality, offering healthcare providers a powerful tool for early disease detection, precise diagnosis, and optimized treatment planning.
With its innovative design and enhanced performance capabilities, the PHAROS scanner is poised to set new standards in neurology. Furthermore, the PHAROS is multi-functional with the ability to physically orient the patient seat and detector configurations for extremity and breast imaging, as well as converting to both lying and seated modes for brain imaging.
The development of PHAROS was supported by the Korea Medical Device Development Fund (KMDF), reflecting a collaborative effort across multiple government agencies to advance innovative medical imaging technologies.
With FDA clearance secured, Brightonix Imaging will begin the process of rolling out the PHAROS PET Scanner to medical facilities across the United States.




