Image source: PR Newswire
South Korea based Daewoong Pharmaceutical (Daewoong) has recently announced the result of its phase 2 clinical trial on enavogliflozin, SGLT2 inhibitor for diabetes currently in the process of development, for the first time at the 2020 International Congress of Diabetes and Metabolism (ICDM) held on Sep. 18-19.
The 2020 ICDM, an international conference sponsored by the Korean Diabetes Association (KDA), was held for the tenth time this year. It was organized as an online event to prevent the spread of COVID-19.
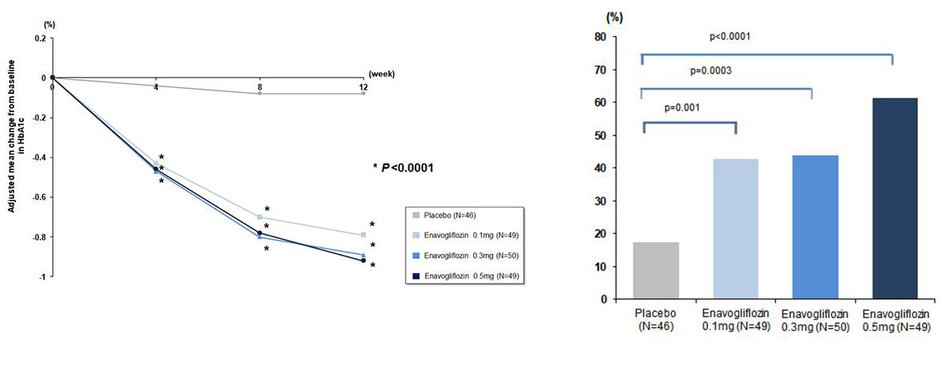
When enavogliflozin was administered for 12 weeks in type-2 diabetic patients with inadequate glycemic control by diet and exercise, the patients showed statistically significant decreases in their glycated hemoglobin (HbA1c) levels in comparison to the placebo starting from week 4. In week 12, the patients' glycated hemoglobin levels decreased by approximately 0.9%p in comparison to the placebo. This is a statistically significant result and indicates an additional decrease in the glycated hemoglobin by approximately 0.2–0.3%p compared to SGLT2 inhibitors of other companies for which trials were conducted in subjects in Western countries. This result, as such, raises expectations for further studies.
The percentage of patients whose glycated hemoglobin level under 7.0%* in week 12 as therapeutic response was as high as 61%, which was an increase by more than 20%p from those in the trials conducted on existing SGLT2 inhibitors. In addition, the percentage of patients achieving the HbA1c reduction at least 0.5% at week 12 was recorded as 72% at most, indicating a notable drug effect to lower serum glucose level, when considering non-responders in other SGLT2 inhibitors.




