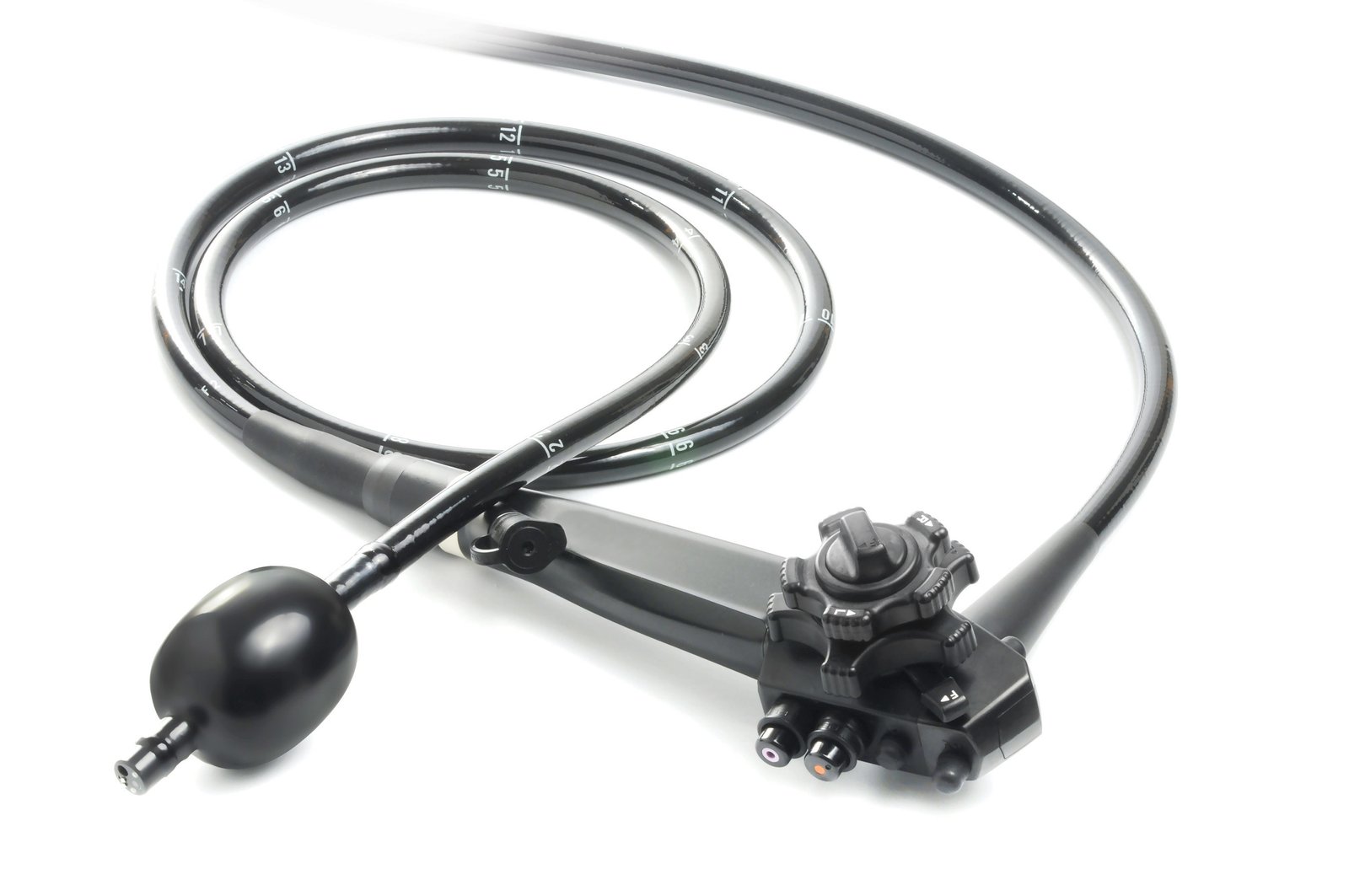
Photo Credit: prnewswire
Israel's SMART Medical Systems Ltd., a developer and manufacturer of innovative endoscopy products, received FDA clearance for its G-EYE® Colonoscope. With this additional FDA clearance, G-EYE® is now available for use in the U.S. market on the commonly used colonoscope models of all three leading endoscopy brands - OLYMPUS, FUJIFILM, and PENTAX Medical.
"The ability to offer G-EYE® on colonoscope brands and models commonly used and widely available in the United States is an important milestone for SMART Medical, patients, and endoscopists," said Gadi Terliuc, Chief Executive Officer of SMART Medical.
"The majority of U.S. endoscopists now have the option to utilize our cutting-edge technology, which has been shown in clinical studies to improve visualization compared with standard colonoscopy, while using their preferred brand and model of colonoscope. We are very excited to have completed our portfolio of U.S. G-EYE® offerings and believe that the widespread availability of the technology on the commonly used colonoscope models has the potential to accelerate adoption of G-EYE® colonoscopy as the standard of care."
The G-EYE® Colonoscope is a 510(k) cleared colonoscope, remanufactured by SMART to include a proprietary balloon at its distal bending section. Withdrawal of the G-EYE® Colonoscope with the balloon moderately inflated during colonoscopy assists in controlling the colonoscope's field of view and positioning. A study demonstrated that G-EYE® can improve colonoscopy outcomes compared with standard colonoscopy across several metrics, including increasing adenoma detection rate (ADR) by 28%, detecting 47% more adenomas per patient (APP), 62% more advanced and large adenomas, and 142% more flat adenomas.




