Photo credit: GlobalData
There is currently 445 chimeric antigen receptor (CAR-T cell) therapies under development by companies headquartered across the Asia-Pacific (APAC) region. With research in progress and increased understanding of cancer biology, the new therapies are expected to make the global market more competitive and cost-effective, says GlobalData, a leading data and analytics company.
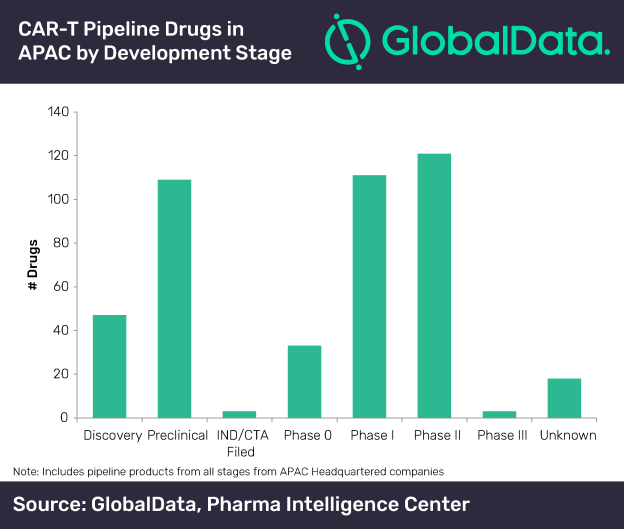
GlobalData’s research reveals that the majority of the cellular modalities are in the early stages of clinical development. Acute lymphocytic leukemia (ALL), B-cell acute lymphocytic leukemia and refractory multiple myeloma are the three major indications for which CAR-T cell therapies are under development in APAC.
Gowri Prasad Gutti, Director of Pharma Intelligence at GlobalData, comments: “Cancer is becoming a serious health concern across the APAC region, because of the ageing population, changes in lifestyle associated with economic development and epidemiologic transition.
“Clinical outcomes of CAR-T cell therapies have been impressive and have shown remarkable results in relapsed/refractory patients. However, the evidence of efficacy is still being gathered with more clinical trials underway.”
In 2019, Japan's Ministry of Health, Labor and Welfare (MHLW) has approved the first CAR-T cell therapy Kymriah (tisagenlecleucel) for the treatment of ALL and diffuse large B-cell lymphoma. The drug received approval for the treatment of Acute Lymphocytic Leukemia (ALL) in Australia in 2018.
According to GlobalData, as on 10 February 2020, there are 369 trials that are ongoing in the APAC region. Most of these trials are in Phase I, followed by Phase I/II.
Gutti explains: “This suggests that the CAR-T modalities are still under clinical evaluation phase and a considerable amount of data evaluation is required for these to be successful as a new generation of anticancer drugs.”
China, by far, leads in the number of CAR-T cell clinical trials, accounting for about 95% of the total clinical trials in the APAC region.
Gutti concludes: “Even though China leads the CAR-T cell therapy development, the approval of CAR-T therapy in China will likely take some time as the majority of the programs are in early stages of development.”




